Together We Can Save Lives
We can prevent overdoses and save lives by ensuring equitable access to essential health care and support services without stigma.
Primary Prevention
Preventing substance use disorder is the first step towards addressing overdoses. Learn about effective prevention programs and safe prescribing practices.
Primary PreventionEvidence-Based Treatment
When a person is ready, high-quality treatment must be available without delay. Help improve access to treatment.
Evidence-Based TreatmentRecovery Support
Recovery support services can lead to better long-term outcomes, especially when available in communities where they are needed. Explore different types of recovery services.
Recovery SupportThe overdose crisis is national, but the impact is personal.
As the overdose crisis continues to change, we must take a pragmatic, evidence-based approach to saving lives, reducing risk, and removing barriers to effective interventions. This requires that we provide care and services that respect the health and dignity of people who use drugs.
Trends in U.S. Drug Overdose Deaths (December 1999–June 2023), by Drug Type*
The overdose crisis has evolved over time and is now largely characterized by deaths involving illicitly manufactured synthetic opioids, including fentanyl and, increasingly, stimulants.
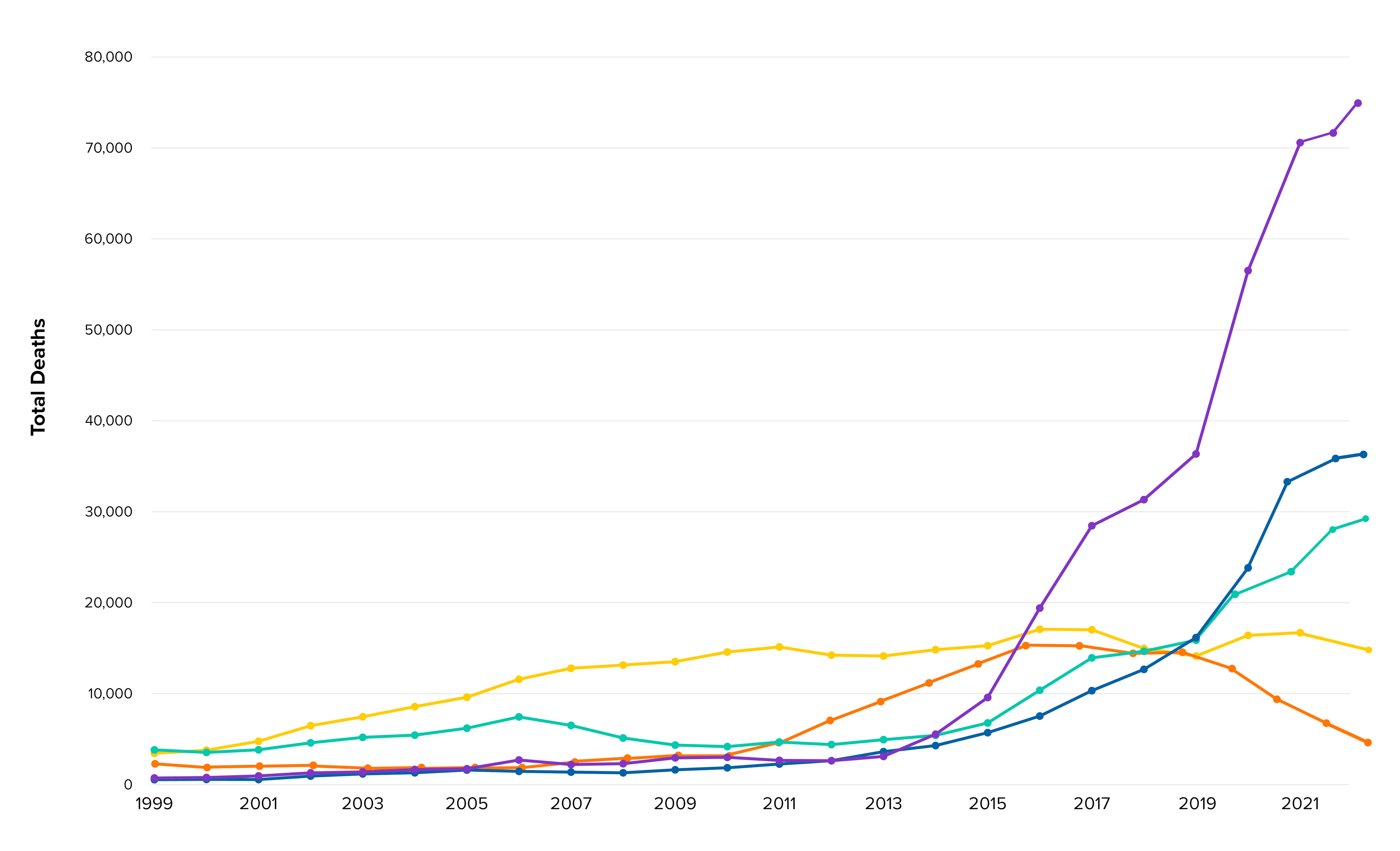
Synthetic opioids excluding methadone overdose deaths increased 103-fold
Psychostimulants with abuse potential (primarily methamphetamine) overdose deaths increased 64-fold
Cocaine overdose deaths increased 7.6-fold
Rx opioid overdose deaths increased 4.1-fold
Heroin overdose deaths increased 2.5-fold
*This graph shows the total number of drug overdose deaths in the United States over the previous 12 months for each month from December 1999 through June 2023, by drug type. Overdose deaths of all intents are included, using underlying cause of death ICD-10 codes X40–X44 (unintentional overdose), X60–X64 (suicide), X85 (homicide), and undetermined intent (Y10–Y14). Drug and drug category involvement are identified by specific multiple cause-of-death codes (heroin: T40.1, prescription opioids: T40.2 and T40.3, synthetic opioids excluding methadone (primarily fentanyl): T40.4, cocaine: T40.5, and psychostimulants with abuse potential (primarily methamphetamine): T43.6. Data source: CDC WONDER Multiple Cause of Death data file (1999–2021: final data file; 2022–2023: provisional data file, accessed 1/24/24). Source: National Vital Statistics System Mortality File
Overdose Deaths Increased
Drug overdose deaths reached 93,331 in 2020 – the highest number ever recorded.
Higher Rates in Minority Groups
In 2019, non-Hispanic American Indians or Alaska Natives had a higher drug overdose death rate than any other racial or ethnic group (30 per 100,000).
Instability Increases Overdose
People without health insurance, or who were incarcerated, or who are living in poverty are at increased risk of fatal opioid overdose.
Working Together to Combat the Overdose Crisis
Canada and the U.S. have published a white paper, Substance Use and Harms During COVID-19 and Approaches to Federal Surveillance and Response, that examines rapid and innovative approaches used by both countries to monitor substance use trends during the pandemic, including:
- substance use harms and deaths in Canada and the U.S.
- the impact of COVID-19 on the opioids crisis
- policy responses to address substance use from the outset of the COVID-19 pandemic to September 2022
